The Mandate for Item-Level Traceability: DSCSA & EU MDR
The global healthcare supply chain is undergoing a significant overhaul, driven by stringent regulations designed to enhance patient safety and combat counterfeiting. In the United States, the Drug Supply Chain Security Act (DSCSA) aims to build an interoperable electronic system to identify and trace certain prescription drugs as they are distributed in the U.S. (U.S. Food & Drug Administration, n.d.). By the final enforcement date (expected 2024-2025), DSCSA mandates package-level serialization, requiring manufacturers, repackagers, wholesale distributors, and dispensers to manage and exchange transaction information, history, and statements electronically. This fundamentally shifts the industry from lot-level to item-level tracking for virtually all prescription drugs. Across the Atlantic, the European Medical Device Regulation (EU MDR) imposes similar, equally rigorous requirements for medical devices (European Commission, n.d.). A cornerstone of EU MDR is the Unique Device Identification (UDI) system, demanding that all medical devices bear a UDI, which must be submitted to the EUDAMED database. This ensures traceability from manufacturing through distribution to the end-user or patient. For supply chain professionals, both regulations present a formidable challenge: efficiently capturing, managing, and exchanging vast amounts of unique product data at an individual item level, a task that traditional methods struggle to scale efficiently. The goal is clear: an accurate, immutable record of every product's journey, making manual, labor-intensive processes obsolete. (EUHub, 2023)

UHF RFID: The Engine of Modern Traceability
In this demanding regulatory landscape, Ultra-High Frequency (UHF) RFID emerges as a pivotal technology for achieving compliance and unlocking significant operational advantages. Unlike conventional 2D barcodes, which require line-of-sight scanning for single items, UHF RFID offers non-line-of-sight and bulk reading capabilities. This means hundreds of items can be identified and authenticated simultaneously, dramatically speeding up processes like serialization, aggregation, and verification—all critical components of DSCSA compliance. For pharmaceutical companies, RFID streamlines the complex task of creating parent-child relationships for aggregated shipments, linking individual drug packages to cases and cases to pallets with unprecedented efficiency. This directly addresses DSCSA's requirement for robust data exchange and verification through an 'interoperable electronic system' (FDAnews, 2023). Similarly, for medical devices under EU MDR, RFID facilitates seamless UDI capture and ensures accurate inventory management, supporting post-market surveillance efforts. Beyond mere compliance, RFID drives substantial operational efficiencies. It automates inventory counts, reducing manual errors and labor costs by up to 80% (Tag N Trak It internal data). Real-time inventory accuracy minimizes waste from expired products, prevents stock-outs, and optimizes supply chain logistics. By embedding unique identifiers directly into the physical flow of goods, RFID creates a 'digital twin' of the supply chain, enabling proactive management and informed decision-making. The ability to read, write, and re-write data on tags further enhances its utility, allowing for dynamic updates throughout a product's lifecycle.

Technical Prowess: RFID's Adaptability in Healthcare
The effectiveness of UHF RFID in diverse healthcare environments hinges on sophisticated technical advancements, particularly in tag design and system integration. Operating in globally standardized frequency bands (e.g., 902-928 MHz in North America, 865-868 MHz in Europe), modern RFID systems utilize tags composed of a microchip (Integrated Circuit) storing the Electronic Product Code (EPC) and an antenna engineered for optimal performance. Crucially, specialized tag types have been developed to overcome the unique challenges posed by pharmaceutical and medical device packaging: * **On-Metal Tags:** Essential for tracking surgical instruments or metal-encased devices, these tags feature insulating layers to prevent signal interference. * **Liquid-Tolerant Tags:** Designed to perform reliably on items with high liquid content, such as vials, syringes, or IV bags, where liquids can otherwise attenuate RFID signals. * **Small Form Factor Tags:** Tiny tags enable serialization of small drug packages or individual medical components. * **Sterilizable Tags:** Engineered to withstand harsh sterilization processes like autoclaving or gamma irradiation, these are vital for reusable surgical instruments. * **Tamper-Evident/Authenticity Tags:** Integrating features that indicate if a package has been opened or if the product is genuine, these tags provide a crucial layer of defense against counterfeiting and diversion. These advanced tags, combined with fixed readers at chokepoints, handheld readers for mobile operations, and tunnel readers for high-speed throughput, create a robust data capture infrastructure. Sophisticated RFID middleware then filters and interprets this raw data, seamlessly integrating it with existing enterprise systems like WMS, ERP, MES, and even Electronic Health Records (EHRs). This end-to-end integration is key to generating the comprehensive, interoperable electronic records required by DSCSA and the UDI data submissions mandated by EU MDR.
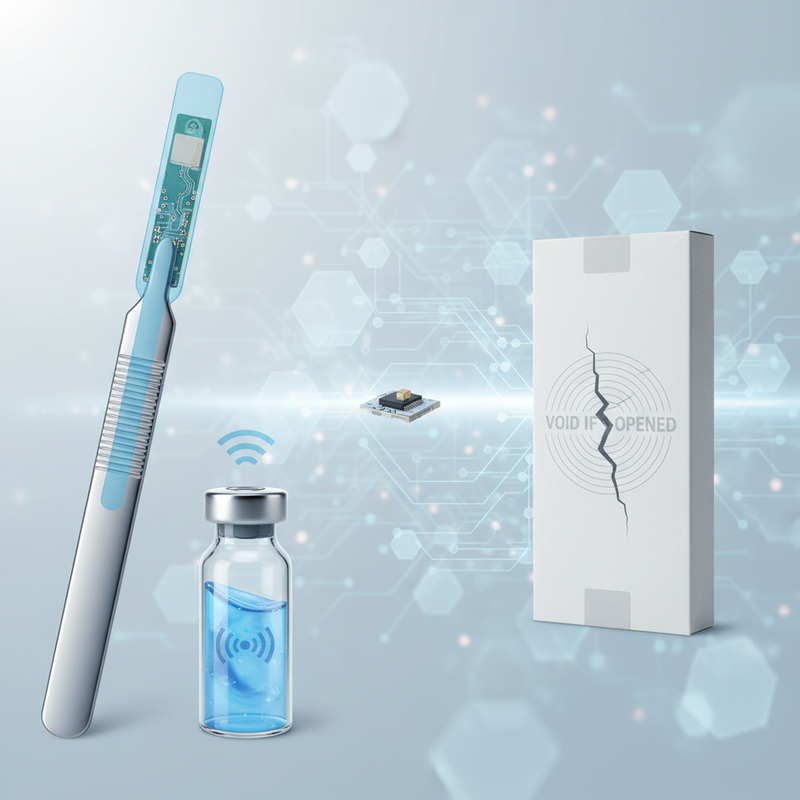
Real-World Impact: RFID in Pharma & Medical Devices
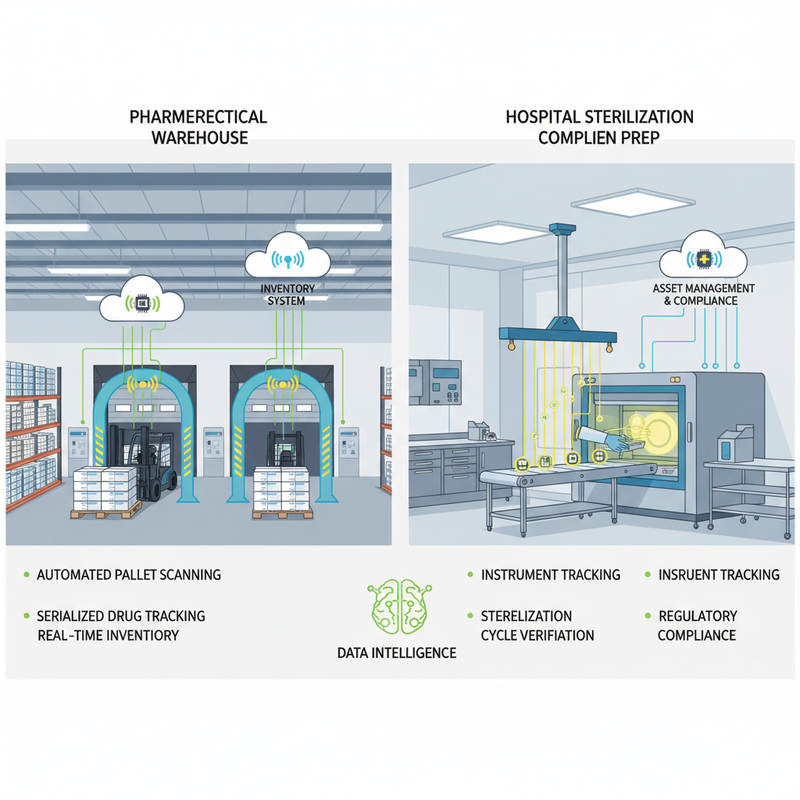
The theoretical advantages of UHF RFID translate into tangible benefits across the pharmaceutical and medical device supply chains, driving both regulatory compliance and operational excellence. **Pharmaceutical Traceability (DSCSA Compliance):** RFID significantly enhances serialization and aggregation processes. Tags on individual drug packages facilitate automated serialization, with these items then aggregated to cases and pallets, all linked via RFID. This dramatically simplifies the creation of parent-child relationships, a complex DSCSA requirement. For instance, many pharmaceutical 3PLs are deploying RFID portals at shipping and receiving docks to automatically verify aggregated shipments, boosting efficiency and reducing manual errors. Companies like **Antares Vision Group** (through its partner networks) have been instrumental in implementing RFID for DSCSA compliance, streamlining aggregation and disaggregation for improved data integrity and speed (Antares Vision Group, n.d.). Beyond aggregation, RFID provides real-time visibility into warehouse inventory, expedites receiving and shipping verification against Advanced Shipping Notices (ASNs), and enables rapid identification and isolation of products during recalls or returns, minimizing patient risk. The integration of secure RFID tags with cryptographic features also offers a powerful deterrent against counterfeit drugs. **Medical Device Traceability (EU MDR & Hospital Operations):** For medical devices, RFID enables automated Unique Device Identification (UDI) capture and management. RFID tags encoding UDI data can be applied to devices, allowing for seamless data submission to EUDAMED and comprehensive internal tracking. Hospitals like **Methodist Hospital (Dallas, TX)** have successfully used RFID to track high-value inventory in surgical services, including implants and consumables, ensuring UDI compliance, reducing waste from expired products, and improving charge capture (RFID Journal articles, various years). Sterilizable RFID tags are revolutionizing surgical instrument tracking, providing audit trails through sterilization cycles, minimizing lost instruments, and ensuring complete surgical kits—critical for infection control and patient safety. Companies such as **Censis Technologies** offer RFID-based solutions that enable hospitals to manage thousands of instruments, comply with cleaning protocols, and prepare accurate surgical trays (Censis Technologies, n.d.). This holistic approach to asset management extends to high-value assets like catheters and stents, preventing diversion, ensuring availability, and optimizing stock levels within hospital pharmacies and nursing stations. Ultimately, RFID fortifies post-market surveillance and recall capabilities, allowing for precise identification of affected devices as required by EU MDR, thereby enhancing patient safety.
Standardization, Growth, and the Future of Healthcare RFID
The widespread adoption and effectiveness of RFID in meeting the stringent demands of DSCSA and EU MDR are deeply rooted in standardization and a rapidly expanding market. The adoption of GS1 standards, particularly EPCglobal Gen2v2 (ISO 18000-6C) and the EPC Tag Data Standard, is fundamental. These standards ensure that data encoded on RFID tags (such as Global Trade Item Numbers - GTINs, Serialized GTINs - SGTINs, and Global Location Numbers - GLNs) is universally recognized and interpretable across the entire supply chain (GS1 US, n.d.). This interoperability is paramount for the seamless data exchange mandated by both regulations. As regulatory deadlines loom and requirements mature, the healthcare RFID market is experiencing significant growth. Analysts project the global healthcare RFID market, valued at approximately **USD 3.0 - 4.5 billion in 2023**, to reach **USD 8.0 - 12.0 billion by 2030**, exhibiting a robust Compound Annual Growth Rate (CAGR) of **15-18%** from 2024 to 2030 (Grand View Research, n.d.; MarketsandMarkets, n.d.). The primary drivers for this expansion are unequivocally regulatory compliance (DSCSA and EU MDR), coupled with a strong desire for improved patient safety, operational efficiency, and advanced asset management. This growth is concentrated across RFID tags, readers, software, and services, with pharmaceutical and medical device tracking segments leading the charge. Key players like Impinj, Zebra Technologies, Avery Dennison, and NXP Semiconductors are at the forefront of hardware innovation, while specialized solution providers such as Terso Solutions and Kit Check are delivering tailored software and smart inventory solutions. The future of healthcare RFID is one of increasing integration, where robust, standardized technology becomes an indispensable backbone for a transparent, secure, and highly efficient global healthcare supply chain.

Conclusion
The journey to DSCSA and EU MDR compliance is not merely a regulatory hurdle but a strategic opportunity to modernize supply chain operations. UHF RFID provides the essential technological foundation, delivering the granular, real-time item-level visibility mandated by these regulations, while simultaneously driving unparalleled operational efficiencies and patient safety improvements. By embracing standardized RFID solutions, pharmaceutical and medical device companies can transform compliance into a competitive advantage. Partner with Tag N Trak It to design and deploy an RFID solution tailored to your specific regulatory and operational needs. Unlock the full potential of your supply chain today.
- RFID Strategies for DSCSA 2025
- EU MDR: RFID Adoption Challenges & Opportunities
- Healthcare Track & Trace
- Drug Supply Chain Security Act (DSCSA)
- Medical Devices
- Healthcare RFID Market Size, Share & Trends Report, 2030
- Healthcare RFID Market - Global Forecast to 2030
- RFID Journal articles (various years)
- Antares Vision Group
- Censis Technologies

